 In nuclear fission events, the nuclei may break into any combination of lighter nuclei, but the most common event is not fission to equal mass nuclei of about mass 120 the most common event (depending on isotope and process) is a slightly unequal fission in which one daughter nucleus has a mass of about 90 to 100 u and the other, the remaining 130 to 140 u. The excess mass Δm = M - Mp is the invariant mass of the energy that is released as photons ( gamma rays) and kinetic energy of the fission fragments, according to the mass-energy equivalence formula, E = mc². The total rest masses of the fission products (Mp) from a single reaction is less than the mass of the original fuel nucleus (M). Nuclear fission of heavy elements produces energy because the specific binding energy (binding energy per mass) of intermediate-mass nuclei with atomic numbers and atomic masses close to 61Ni and 56Fe is greater than the specific binding energy of very heavy nuclei, so that energy is released when heavy nuclei are broken apart. The energy of nuclear fission is released as kinetic energy of the fission products and fragments, and as electromagnetic radiation in the form of gamma rays in a nuclear reactor, the energy is converted to heat as the particles and gamma rays collide with the atoms that make up the reactor and its working fluid, usually water or occasionally heavy water. By contrast, most chemical oxidation reactions (such as burning coal or TNT) release at most a few eV per event, so nuclear fuel contains at least ten million times more usable energy than does chemical fuel. Typical fission events release several hundred million eV of energy for each fission event. In a nuclear reactor or nuclear weapon, most fission events are induced by bombardment with another particle, such as a neutron. 
Most nuclear fuels undergo spontaneous fission only very slowly, decaying mainly via an alpha/ beta decay chain over periods of millennia to eons. These fuels break apart into a range of chemical elements with atomic masses near 100 (fission products). The most common nuclear fuels are 235U (the isotope of uranium with an atomic mass of 235 and of use in nuclear reactors, 238) and 239Pu (the isotope of plutonium with an atomic mass of 239). Chemical isotopes that can sustain a fission chain reaction are called nuclear fuels, and are said to be fissile. 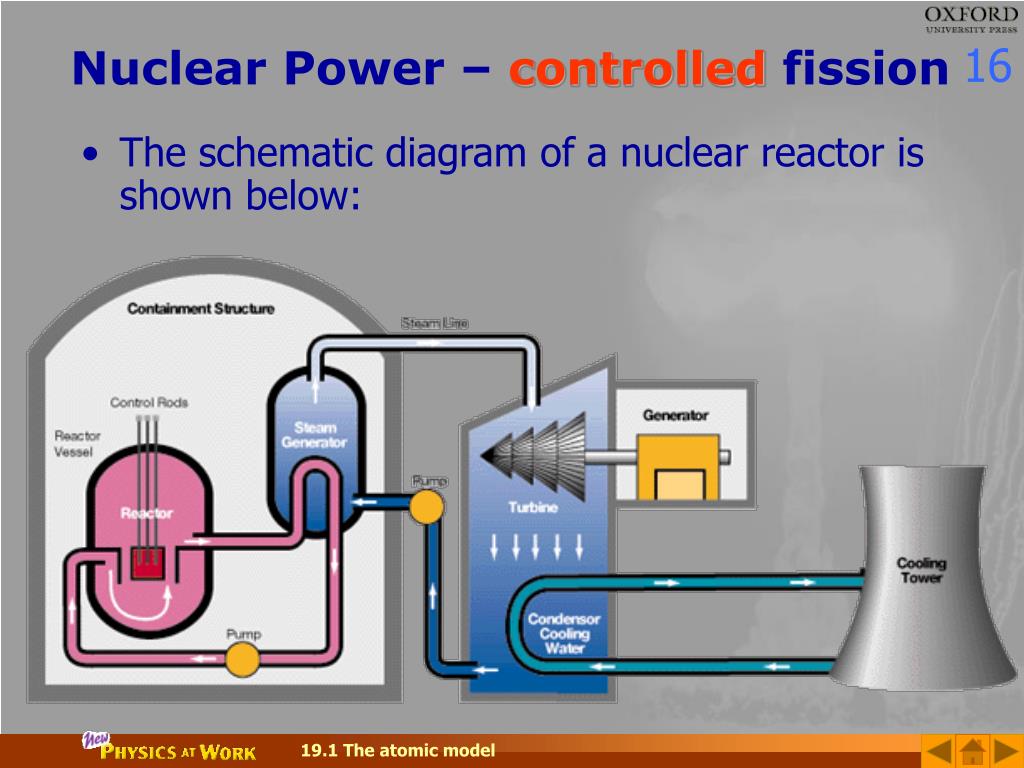
#Which reaction takes place in a nuclear fission reactor free#Nuclear fission differs from other forms of radioactive decay in that it can be harnessed and controlled via a chain reaction: Free neutrons released by each fission event can trigger yet more events, which in turn release more neutrons and cause more fissions. A slow-moving neutron is absorbed by the nucleus of a uranium-235 atom, which in turn splits into fast-moving lighter elements (fission products) and free neutrons. This makes possible a self-sustaining chain reaction that releases energy at a controlled rate in a nuclear reactor or at a very rapid uncontrolled rate in a nuclear weapon.Īn induced nuclear fission event. Both uses are made possible because certain substances, called nuclear fuels, undergo fission when struck by free neutrons and in turn generate neutrons when they break apart. 
Nuclear fission produces energy for nuclear power and to drive the explosion of nuclear weapons. Fission is a form of elemental transmutation because the resulting fragments are not the same element as the original atom. Fission of heavy elements is an exothermic reaction which can release large amounts of energy both as electromagnetic radiation and as kinetic energy of the fragments ( heating the bulk material where fission takes place). Nuclear fission is the splitting of the nucleus of an atom into parts (lighter nuclei), often producing photons (in the form of gamma rays), free neutrons, and other subatomic particles as by-products. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
